sur DBV TECHNOLOGIES (EPA:DBV)
DBV Technologies Unveils Promising Phase 3 VITESSE Study Results at AAAAI 2026
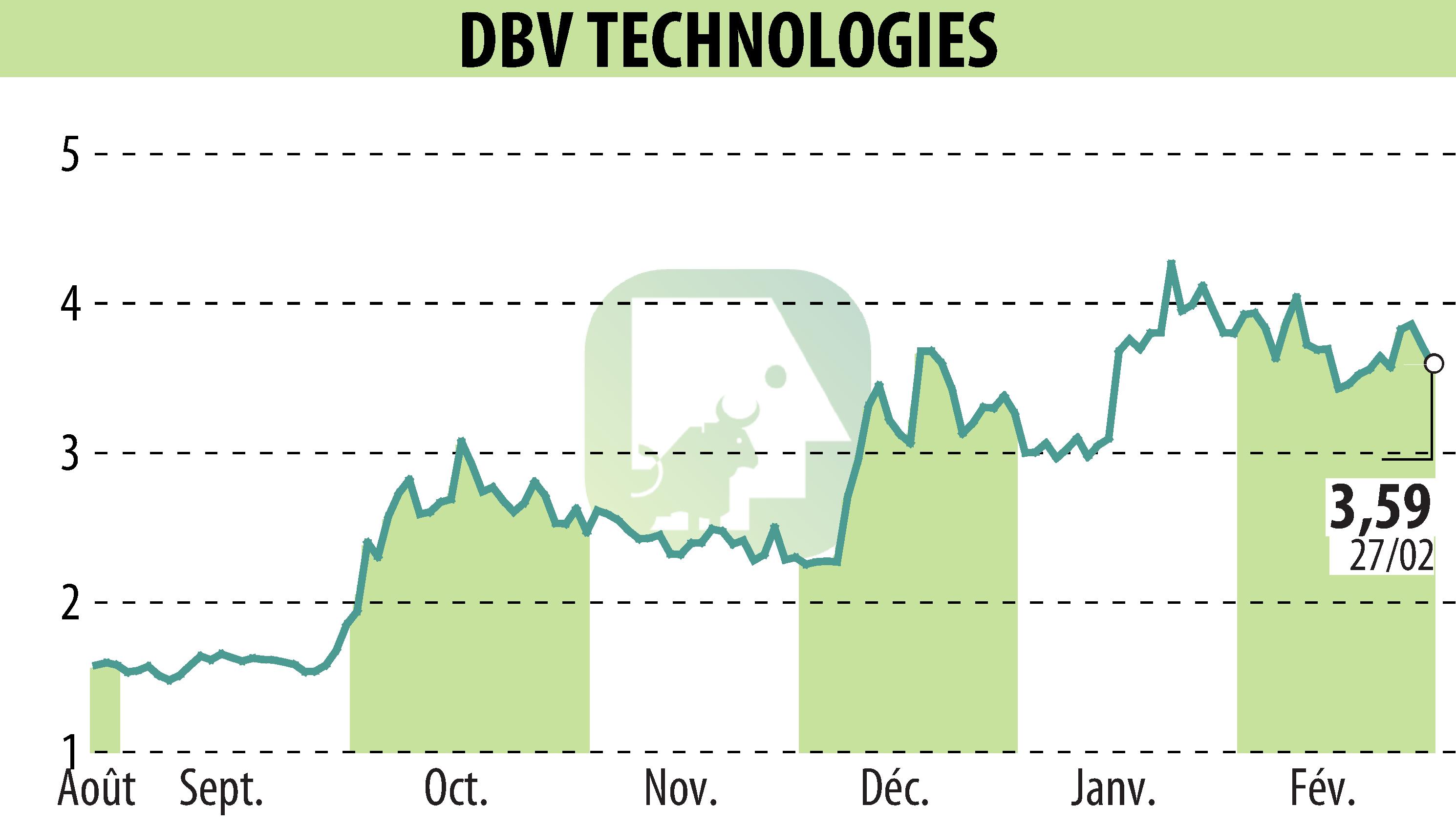
DBV Technologies presented positive outcomes from its Phase 3 VITESSE study at the AAAAI 2026 Annual Meeting. The study focused on the VIASKIN® Peanut Patch for children with peanut allergies, aged 4 to 7. Results showed that 83% of the treated children increased their eliciting dose after 12 months, compared to 48% in the placebo group. Moreover, 60% witnessed an increase of at least two doses, contrasting with 23% in the placebo group.
The study achieved its primary endpoint with significant results: 46.6% of children on the VIASKIN Peanut Patch responded to the treatment versus 14.8% on placebo. This substantial difference confirms the treatment's potential efficacy. The patch was well-tolerated, primarily causing mild local reactions. DBV plans to submit a Biologics License Application to the FDA.
R. P.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de DBV TECHNOLOGIES
