sur DIAGNOSTIC MEDICAL (EPA:ALDMS)
European MDR certification for DMS Group
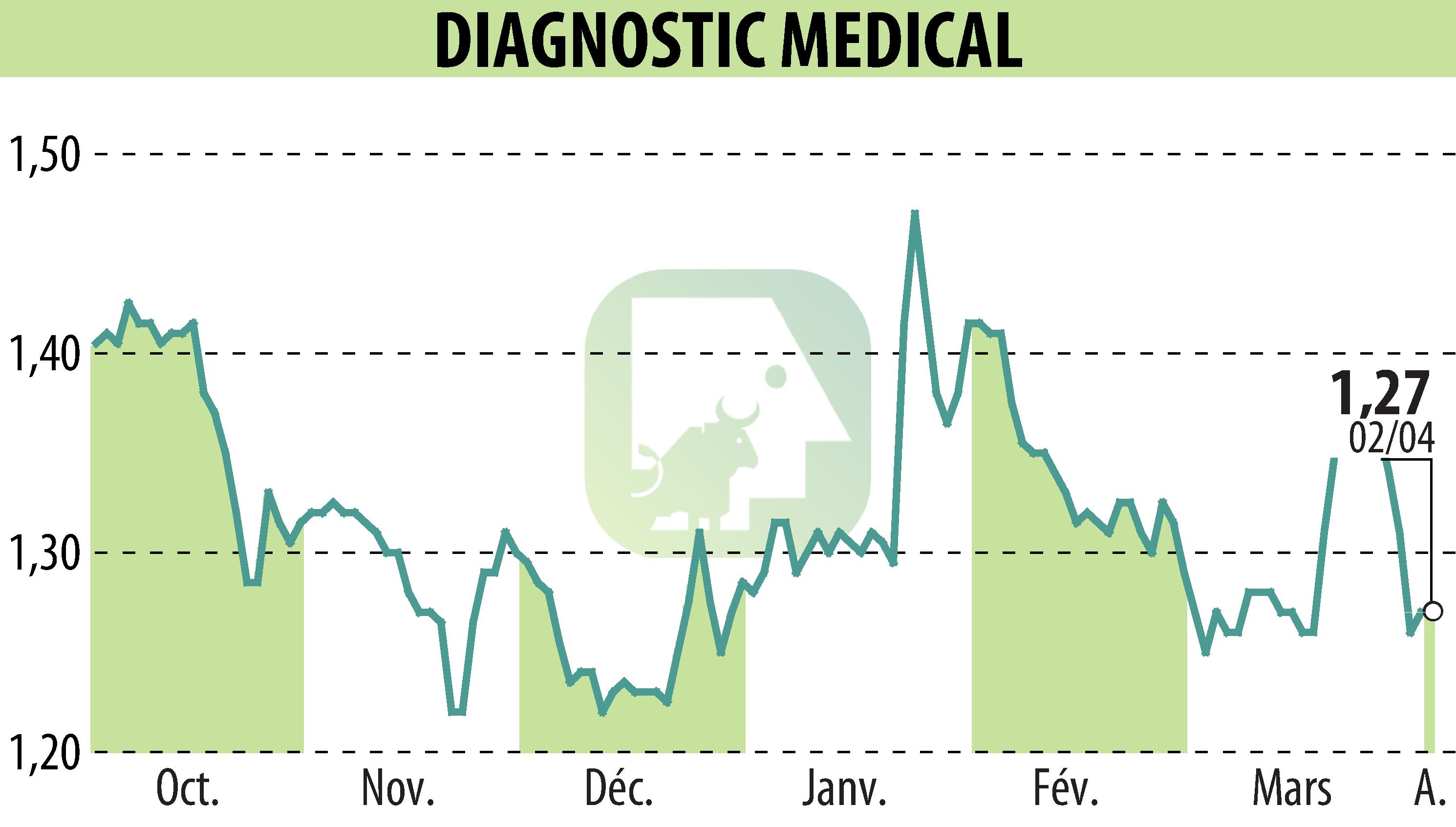
On April 7, 2026, Diagnostic Medical Systems announced that it had obtained MDR certification for its STRATOS range of bone densitometers. This European regulation, in effect since replacing the previous directives, guarantees the conformity of medical devices in terms of safety and quality.
DMS Group, a European leader in this sector, distinguishes itself through its solutions manufactured in France. Certification strengthens its strategic position in the European market. In 2025, the group recorded 8% growth in this area, with a turnover of €11 million.
In addition to this progress, DMS Group is preparing its entry into the US market by submitting an application to the FDA. This market represents 40% of the global bone densitometry market.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de DIAGNOSTIC MEDICAL
