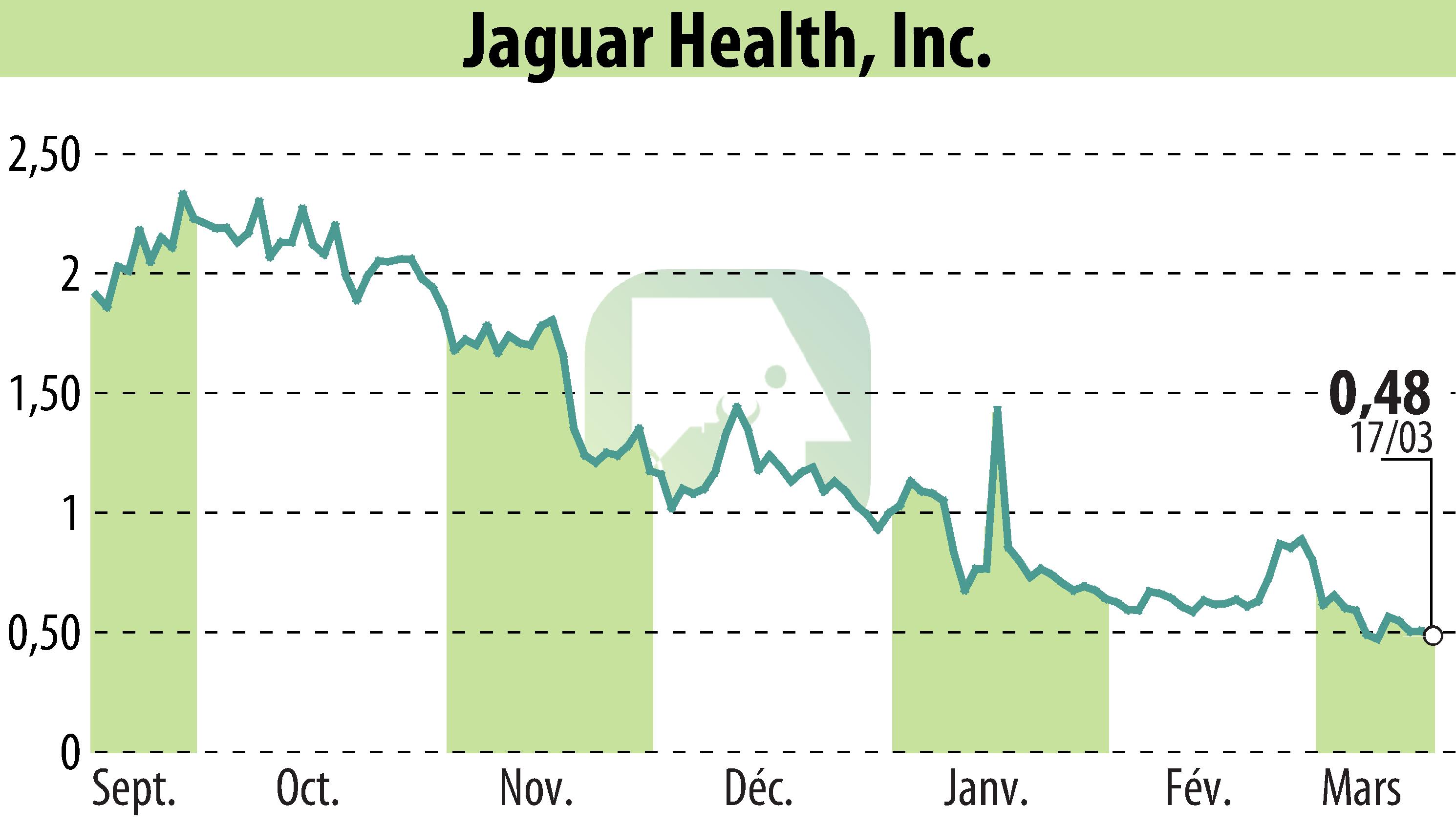
sur Jaguar Health, Inc. (NASDAQ:JAGX)
FDA Supports Extension of Jaguar Health's Crofelemer Trial for MVID
Jaguar Health's subsidiary, Napo Pharmaceuticals, has announced FDA support for an extended trial phase of crofelemer. This trial focuses on pediatric patients with Microvillus Inclusion Disease (MVID), a rare and lethal condition. The trial aims to evaluate the safety and efficacy of crofelemer and its potential to reduce parenteral support needs, which are vital for MVID patients but associated with significant toxicities.
This single-blind extension phase follows a double-blind treatment already completed by 30% of participants. Approvals from various health agencies, including the FDA and European counterparts, back this initiative. The trial's continuation reflects a hopeful step towards new treatments for this orphan disease, possibly leading to expedited drug approval pathways.
Jaguar Health's CEO, Lisa Conte, emphasizes the importance of new therapies for MVID due to its severe nature and lack of existing treatments. The ongoing trial is a collaborative effort with international clinical sites, aiming for potential breakthroughs in treating this ultrarare disorder.
R. H.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de Jaguar Health, Inc.
