sur Nanohale AG (ETR:FYB)
Formycon Secures Licensing for Biosimilar in Europe and Beyond
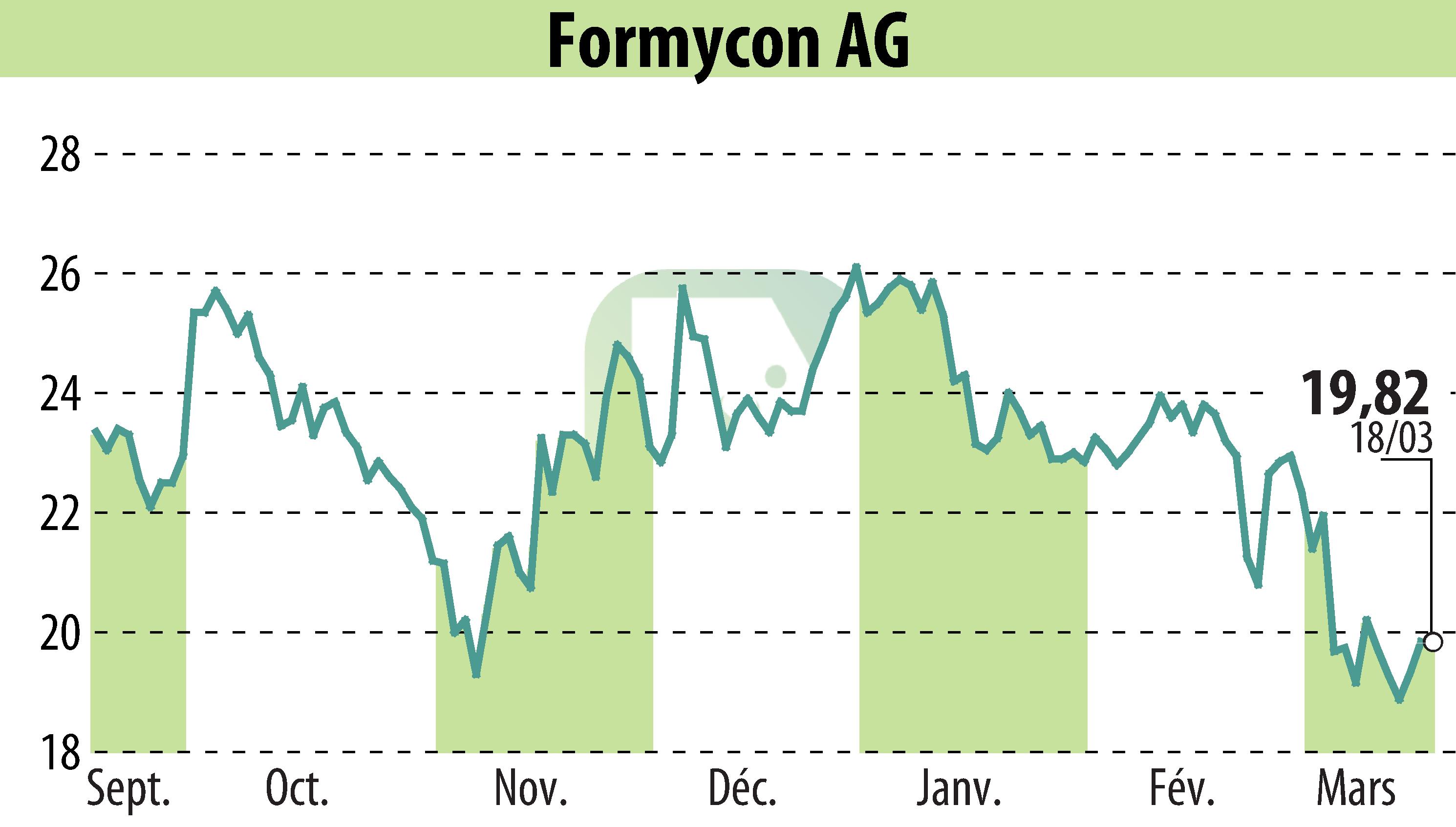
Formycon AG has finalized a settlement and licensing agreement with Regeneron Pharmaceuticals and Bayer Healthcare LLC for its biosimilar FYB203, also known as AHZANTIVE® and Baiama®. This agreement allows the release of these biosimilars in Europe and key markets in Latin America and the Asia-Pacific region starting in May 2026. This follows the October 2025 securing of the U.S. license date, planned for Q4 2026.
AHZANTIVE® and Baiama® target neovascular age-related macular degeneration and other serious retinal diseases, using aflibercept to inhibit the growth of damaging blood vessels in the retina. The settlement resolves all patent litigations, facilitating market access for commercialization partners. For Formycon, this represents a strategic expansion and opportunity for sustainable success in international markets, offering a cost-effective treatment option for retinal diseases.
R. H.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de Nanohale AG
