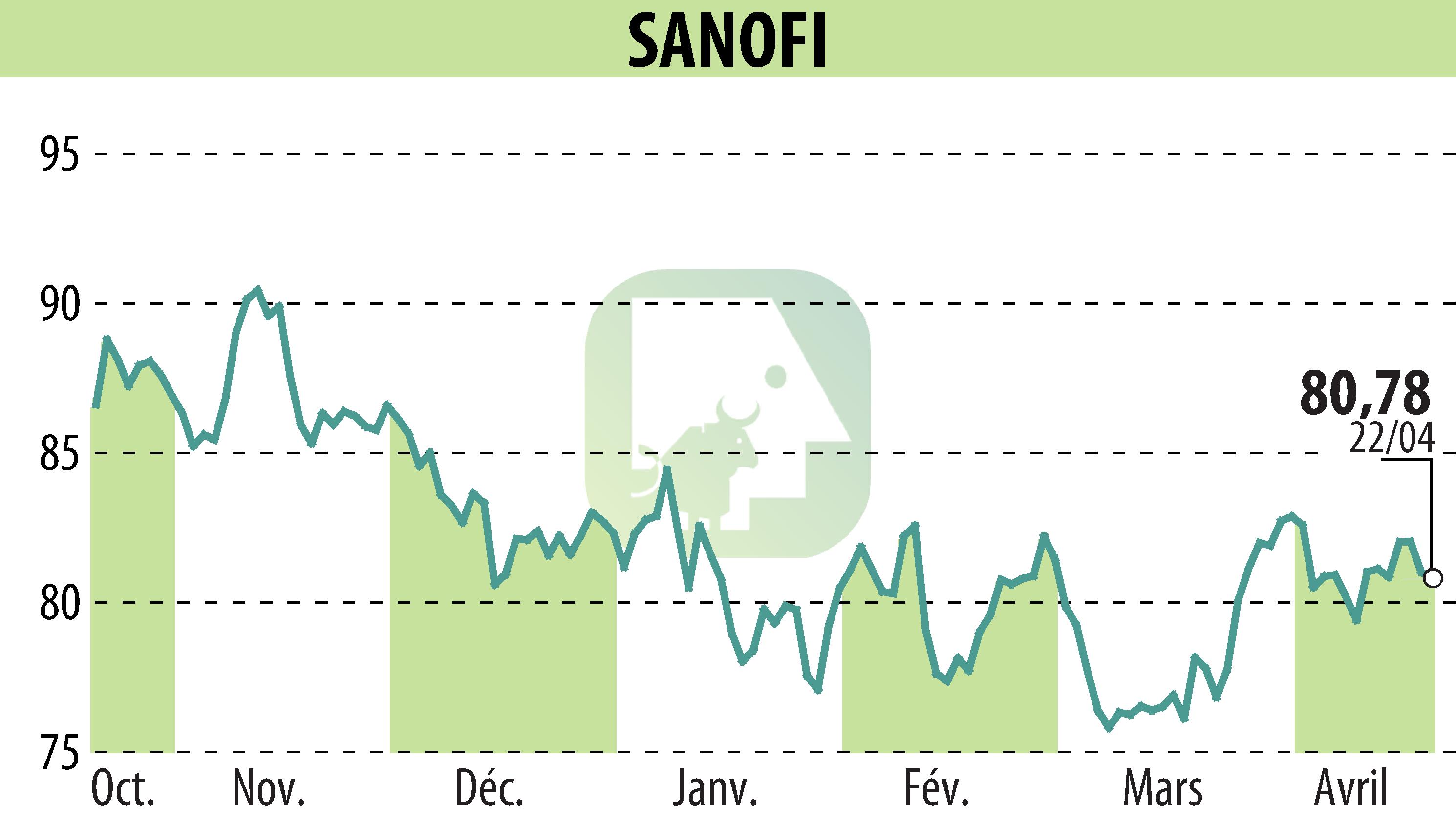
sur SANOFI-AVENTIS (EPA:SAN)
Sanofi and Regeneron Win FDA Approval for Dupixent in Young Children
The US FDA has approved Dupixent, a biologic medicine from Sanofi and Regeneron, for children aged two to 11 with chronic spontaneous urticaria (CSU) not controlled by antihistamines. Dupixent is the first biologic option for this age group.
CSU affects children's quality of life, causing severe itch and hives. Existing CSU treatments are limited, leaving many without sufficient relief. Dupixent is already approved for five type 2 inflammation diseases in children under 12, marking its importance in treating related conditions.
FDA approval is based on positive results from the LIBERTY-CUPID clinical study program, demonstrating significant reductions in itch and urticaria symptoms compared to placebo. Safety data aligns with Dupixent's established profile.
Besides the US, Dupixent has similarly gained approval in the EU and other countries for pediatric CSU cases.
R. H.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
