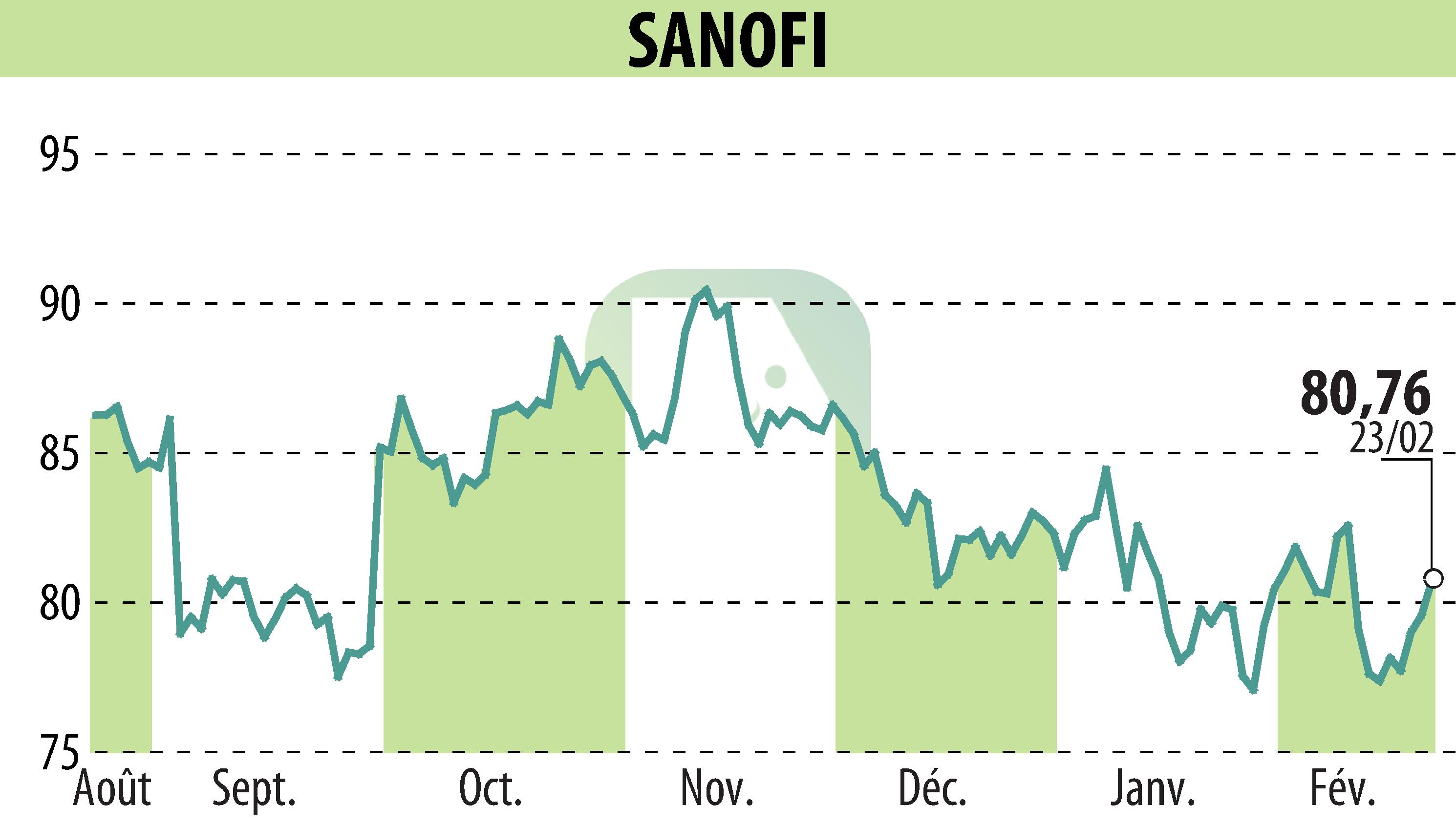
sur SANOFI-AVENTIS (EPA:SAN)
Sanofi and Regeneron's Dupixent Gains FDA Approval for AFRS Treatment
Sanofi and Regeneron have announced that Dupixent (dupilumab) is now approved by the FDA for treating allergic fungal rhinosinusitis (AFRS) in individuals aged six and older. This marks Dupixent as the first medicine approved for AFRS, a type 2 inflammatory disease of the sinuses, often requiring surgery with high recurrence rates. The approval follows a phase 3 study showing significant improvements in sinus opacification, nasal congestion, and reduced need for corticosteroids or surgery.
AFRS is prevalent in warm, humid climates where fungal spores thrive, leading to nasal polyps and serious complications like bone erosion. Dupixent's unique ability to reduce symptoms by targeting IL4 and IL13 pathways sets a new standard of care for AFRS patients. The drug is administered via subcutaneous injection based on age and weight, offering a less burdensome treatment alternative to surgery and extended steroid use.
This FDA approval reinforces Dupixent's position as a versatile medication for a wide range of diseases involving type 2 inflammation. Sanofi and Regeneron plan to seek additional global approvals to broaden patient access.
R. P.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
