sur SANOFI-AVENTIS (EPA:SAN)
Sanofi's Lunsekimig Shows Promise in Asthma and CRSwNP Studies
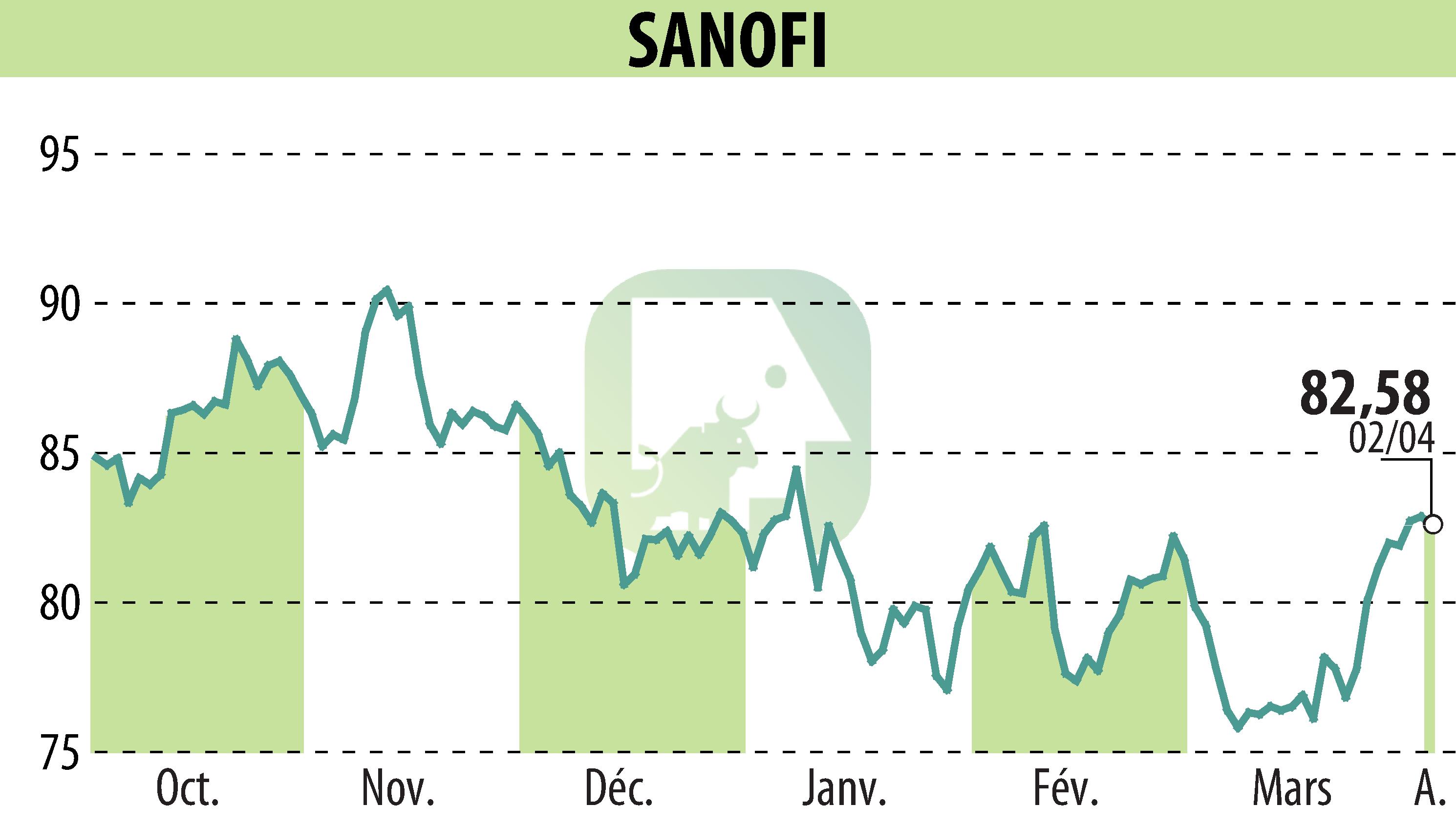
Sanofi's recent phase 2 studies reveal promising results for Lunsekimig in treating chronic respiratory conditions. The AIRCULES phase 2b trial successfully met its primary and secondary objectives in moderate-to-severe asthma, highlighting a significant reduction in exacerbations and lung function improvement. Similarly, the DUET phase 2a study achieved its targets in chronic rhinosinusitis with nasal polyps (CRSwNP), demonstrating positive changes in nasal polyps and congestion scores. However, the VELVET phase 2b study did not achieve its primary goal in atopic dermatitis.
Lunsekimig, a novel bispecific Nanobody® VHH, blocks TSLP and IL-13, key factors in respiratory inflammation. Across all studies, the drug was generally well tolerated. Detailed findings will be presented at future medical congresses. These developments emphasize Lunsekimig's potential role in managing respiratory diseases, although regulatory evaluations remain pending.
R. P.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
