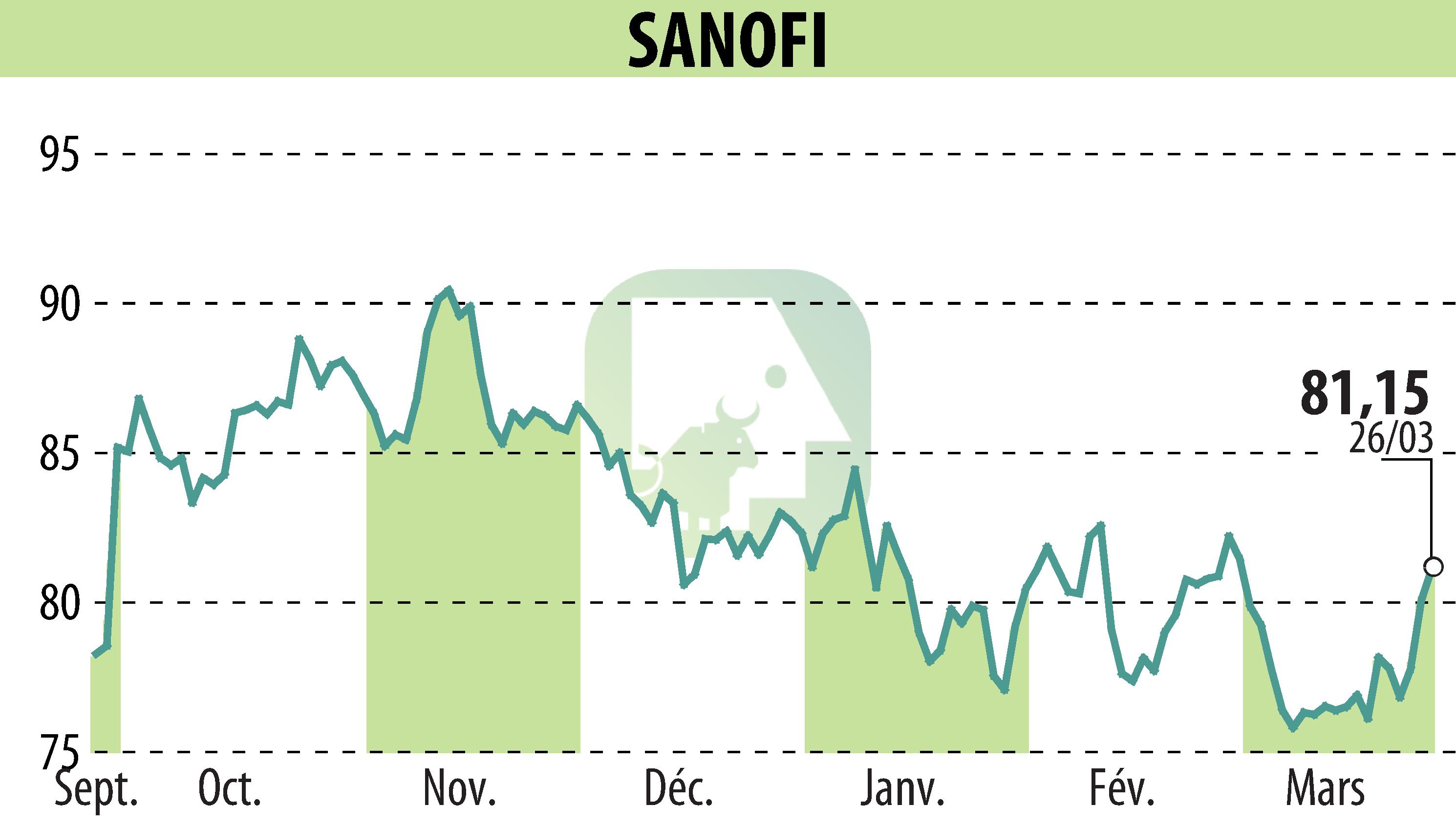
sur SANOFI-AVENTIS (EPA:SAN)
Sanofi's Sarclisa SC Recommended for EU Approval
Sanofi's Sarclisa subcutaneous (SC) formulation, administered via an on-body injector, has been recommended for EU approval by the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP). The recommendation is based on positive results showcasing the efficacy, pharmacokinetics, and safety of the SC formulation compared to the intravenous version.
If approved, Sarclisa SC will be the first anticancer treatment available through an on-body injector and as a manual injection in the EU. The CHMP's positive opinion follows findings from the IRAKLIA phase 3 study, demonstrating non-inferiority of the SC formulation, backed by four additional studies.
The studies utilized Enable Injections’ enFuse® injector, designed to deliver high-volume medications subcutaneously, potentially improving patient comfort and satisfaction. Sarclisa is currently approved in various countries for multiple myeloma treatment in several formulations.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
