sur SENSORION (EPA:ALSEN)
Sensorion continues its development in gene therapy for deafness
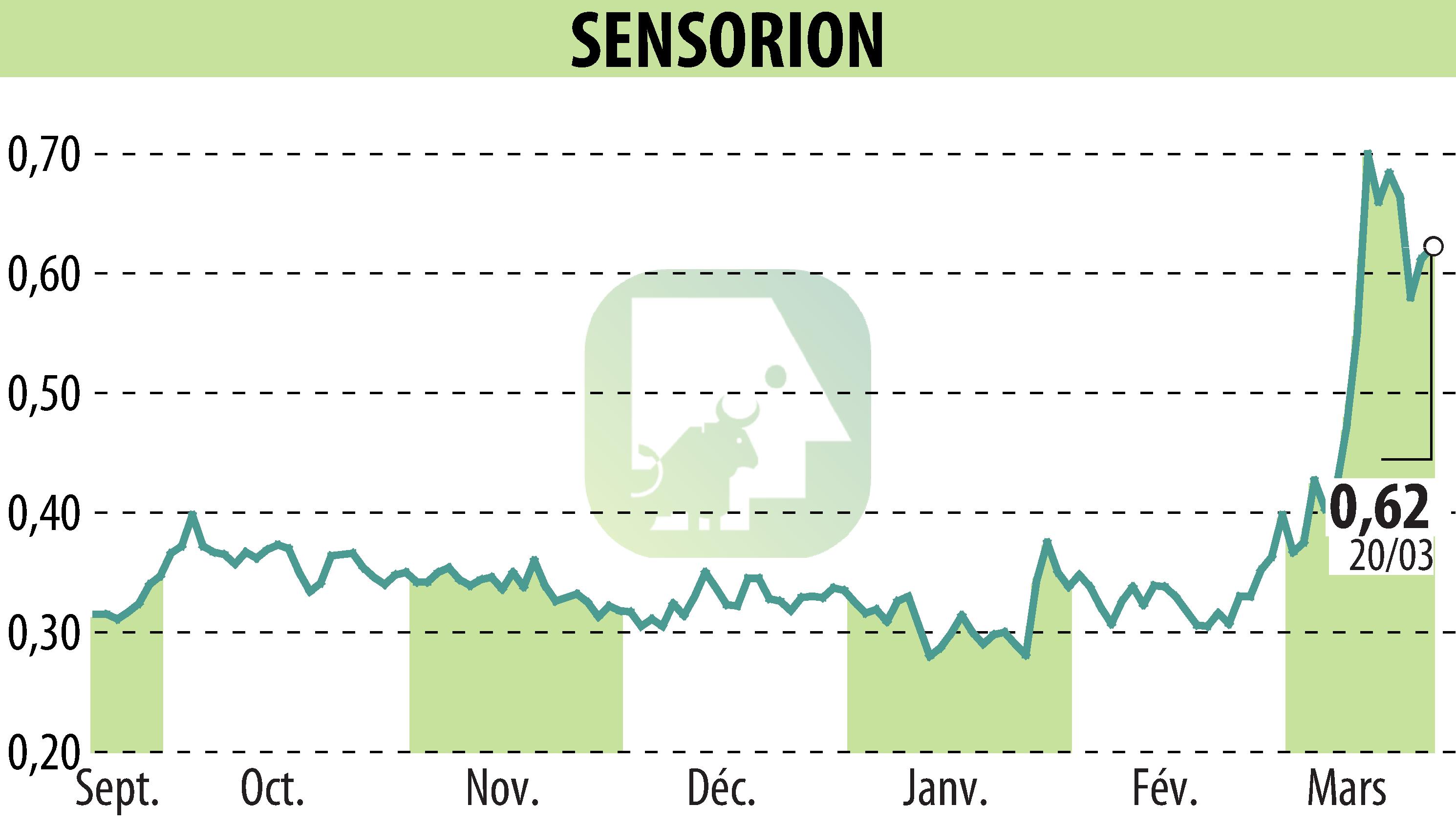
Sensorion has published the six-month results of the Phase 1/2 Audiogene trial of its gene therapy SENS-501. This study targets congenital deafness linked to mutations in the OTOF gene. The data show early signs of efficacy in Cohort 2, suggesting a dose-response relationship. A third dose level is being considered, and consultations with regulatory authorities are planned.
In parallel, Sensorion is making progress on the GJB2-GT (SENS-601) program, which targets deafness linked to the GJB2 gene, the leading cause of congenital deafness. Submission of the CTA is expected in the first half of 2026, and that of the IND by the end of 2026.
Clinical efforts and acquired experience strengthen Sensorion's position in therapeutic innovation for genetic hearing loss, particularly affecting children. This development is supported by strategic collaborations, notably with the Pasteur Institute.
R. P.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SENSORION
